Cellular Rhythms in Fungal Fusion: How Neurospora crassa Germlings Signal and Synchronize
Article audio
Listen to a generated narration of this post.
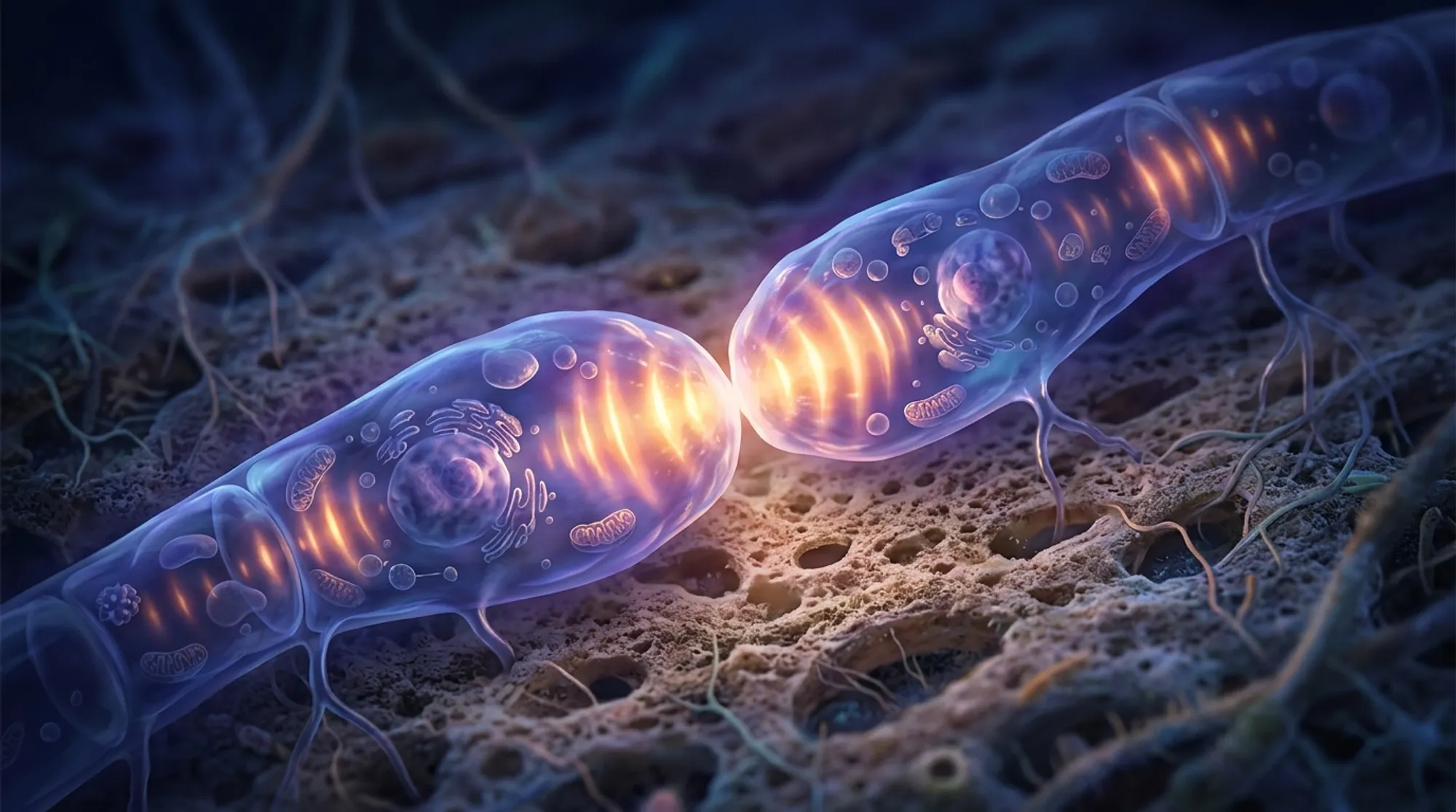
Did you know fungal cells have their own rhythmic ‘conversations’ before they fuse? In the filamentous fungus Neurospora crassa, genetically identical spores don’t just randomly merge—they engage in a carefully coordinated dialog involving pulsed signals that guide them toward one another. This cellular communication, orchestrated by oscillating proteins at the growing tips of germinating spores, sets the stage for fusion and colony formation.
TL;DR
- Researchers identified EOP-1, a protein that oscillates rhythmically at fungal germling tips even when cells are isolated, revealing an intrinsic cellular signaling rhythm.
- These oscillations synchronize between neighboring germlings, initiating a dialog that leads to chemotropic growth and somatic cell fusion, essential for fungal colony development.
Cell fusion is a fundamental biological process that enables cells to merge their contents and functions, playing critical roles in development and tissue formation across eukaryotes. In filamentous fungi like Neurospora crassa, fusion occurs early when germinating spores grow toward each other and merge, forming a connected network called the mycelium. This network is further reinforced by fusion events between hyphal branches within mature colonies. Previous studies revealed key regulators such as the MAP kinases MAK-1 and MAK-2 and the fungal-specific SO protein, which dynamically shuttle between the tips of interacting cells, suggesting a dialog-like communication. However, how this dialog begins—how cells initiate the signaling that leads to fusion—remained unclear.
To uncover the molecular players involved in initiating fungal cell fusion, the researchers focused on SO, a known fusion factor, and searched for its interaction partners using co-immunoprecipitation combined with mass spectrometry. They identified EOP-1, a previously uncharacterized protein, as a direct interactor of SO through biochemical assays and yeast two-hybrid experiments. Using gene deletion mutants, they assessed the role of EOP-1 in fungal growth and fusion. Live-cell fluorescence microscopy allowed visualization of EOP-1 dynamics in germlings, revealing its oscillatory recruitment to cell tips. The team also investigated how EOP-1’s behavior depended on other fusion-related proteins and signaling pathways, including MAK-1 and MAK-2.
The study found that deleting the eop-1 gene abolished germling fusion and chemotropic interactions, confirming EOP-1’s essential role in cell-cell communication. Live imaging showed that EOP-1 oscillates at the tips of interacting germlings in synchrony with SO, supporting the idea of a coordinated signaling dialog. Remarkably, EOP-1 also exhibited similar oscillations in isolated, non-interacting germlings, indicating that each fungal spore has its own intrinsic signaling rhythm. These autonomous oscillations likely serve as pulsed signals emitted in search of fusion partners. The oscillatory recruitment of EOP-1 required the presence of other fusion machinery components, and loss of EOP-1 reduced activation of the MAK-1 pathway, placing EOP-1 upstream in the signaling cascade. Together, these findings reveal a novel mechanism by which fungal cells initiate and coordinate the communication that precedes fusion.
This research provides new insight into the earliest steps of fungal cell fusion, a process vital for colony establishment and growth. Discovering that fungal germlings possess intrinsic oscillatory signaling rhythms that synchronize upon contact advances our understanding of how genetically identical cells initiate communication and coordinate complex behaviors. Beyond fungal biology, these findings contribute to the broader study of cellular communication and signaling dynamics. The visual nature of oscillating protein recruitment at cell tips offers compelling opportunities for microscopy and modeling, potentially informing synthetic biology and biotechnology applications involving fungal systems.
While the study robustly demonstrates EOP-1’s role in initiating fusion-related signaling oscillations, the precise molecular nature of the signals exchanged between cells remains unknown. The identity of the chemoattractant or receptor involved in this dialog has yet to be identified. Additionally, although the oscillations suggest a vesicle-mediated pulsed release mechanism, direct evidence for the vesicles or signaling molecules is still lacking. Future work is needed to elucidate these aspects and to explore how widespread such intrinsic oscillatory signaling may be across other fungal species or cell types.

